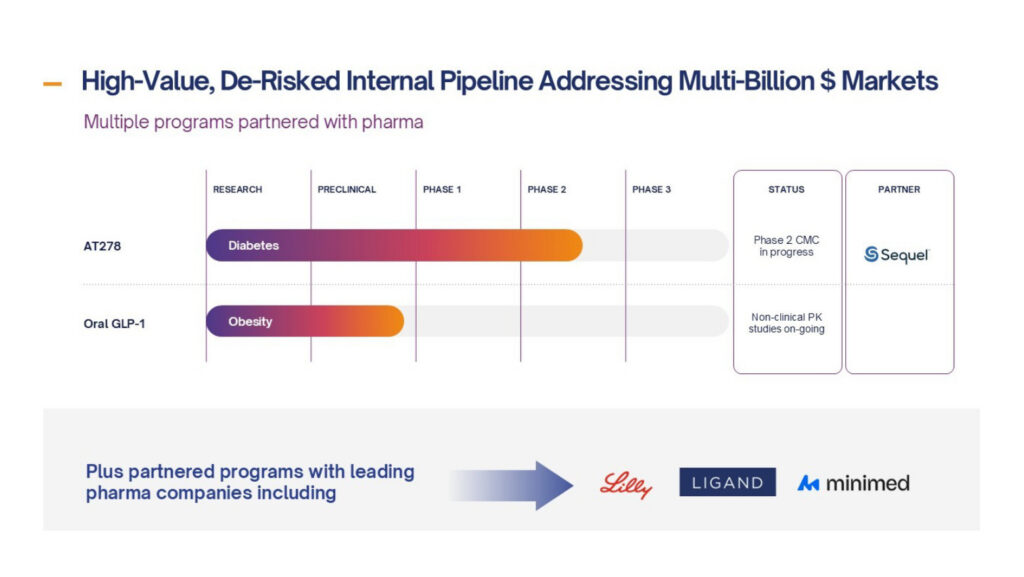
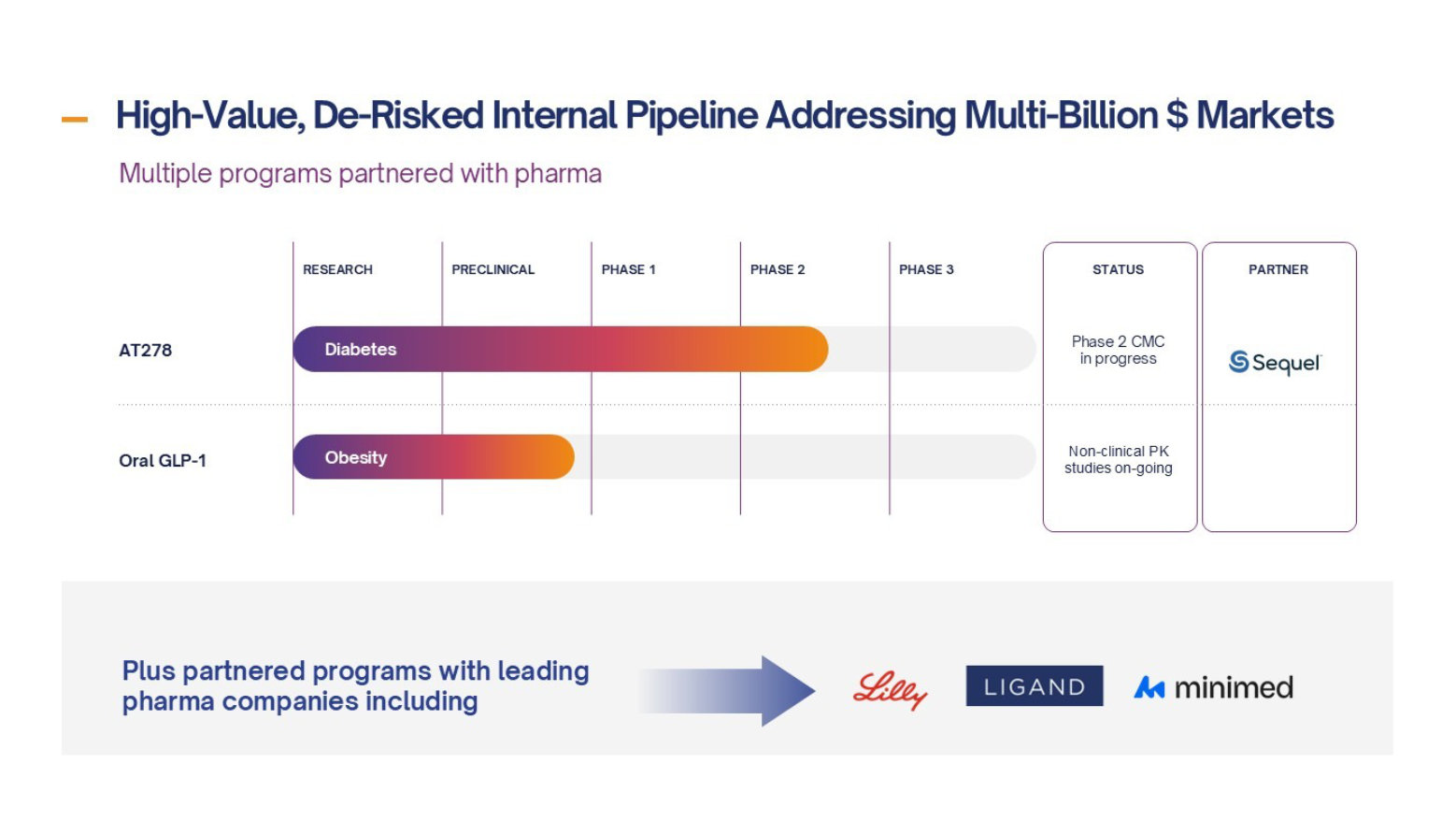
Diabetes
AT278 – a disruptor insulin designed to transform Automated Insulin Delivery (AID) systems with the aim of improving patient care for people living with diabetes, by reducing patient burden and improving outcomes
- The only ultra-concentrated (500U/mL) and ultra-rapid-acting insulin in development
- Unique profile, the only insulin that can enable AID use for high insulin users and drive the next generation of miniaturised, longer wear AID systems
- Best-in-class profile compared to the best insulins available to patients today
- Co-development agreement with Sequel Med Tech for preparatory work for Phase 2 clinical trial of AT278 in their AID system